[Deprecated] CDAGenerator
Gazelle tools documentation is now maintained at https://gazelle.ihe.net/gazelle-documentation/
Introduction
This application has multiple purposes.
- it can be used to generate sample CDA documents that can be used for testing purposes
- today it allows to generate a dispensation document based on a prescription in the context of the epSOS project.
- it will be extended to allow the generation of other kind of CDA documents.
- It hosts the models of the CDA documents and offers the documentation and the edition interface for those models.
- Documentation of the model based contraints
- Generation and Edition of basic CDA R2 documents
Please email questions to the IHE Europe Connectathon Manager (Eric Poiseau) for guidance through the process.

CDA Documentation
CDAGenerator offer a documentation of CDA templates, CDA constraints, and CDA structure.
CDA Structure
CDAGenerator offers a complete documentation of the structure of CDA standard. It is the same description that we find in the schema of CDA R2. The documentation is generated from the model UML that describes the CDA standard. To go to documentation you have to go to menu -> CDA Documentation ->
The html page that describe the Root element (ClinicalDocument) looks like this :

CDA Templates Documentation
All the templates described and validated by CDAGenerator are documented by the tool.
To go to the documentation you have to go to : menu -> Documentation ->
The documentation of the templates is generated automatically from the model UML, which describes the template and its constraints.
CDA constraints documentation
The CDA constraint documentation is a page that contains all the constraint validated by CDAGenerator. This page allows to do search of a constraint by its name, its dependency to a class or a package, or by keyword from the description, etc.
To go to the page of constraint documentation ou have to go to the menu -> Documentation ->
CDA Edition
CDAGenerator offers the possibility to edit and to create basic CDA documents. To do so, you have to go to menu -> CDA Generation ->
You can Generate an empty file, or you can uplaod a CDA file :

To use the module of edition, you have to click on the button edit.
A new page will be opened containg the possibility to edit element by element each sub element of the root ClinicalDocument.

The same page allows to download the modified CDA document, or to validate it, or to save it.
The editor of CDA document is generated also from the model of description of the structure of CDA document.
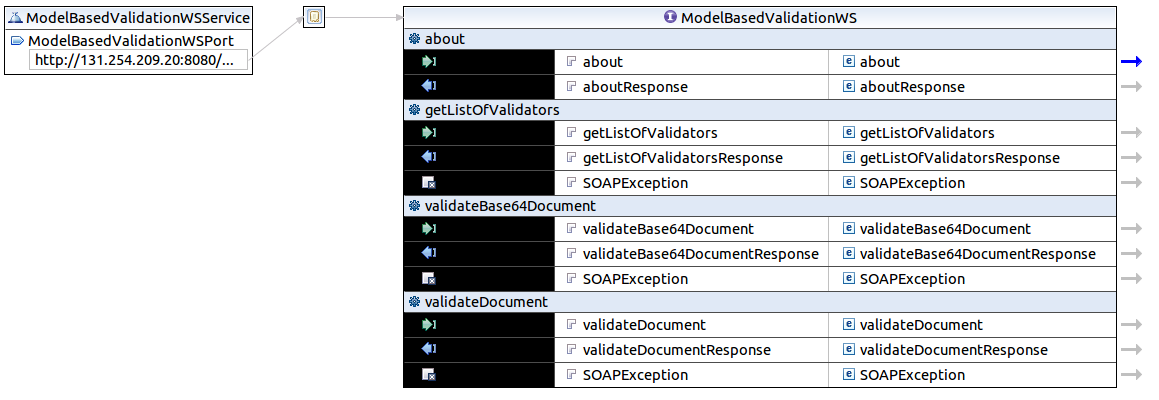
CDA Web service
CDAGenerator include a web service of validation, that could be used as a standalone without using EVSClient, to validate a CDA document
Disclaimer
The CDA Generator Application is an experimental system.
IHE-Europe assumes no responsibility whatsoever for its use by other parties, and makes no guarantees, expressed or implied, about its quality, reliability, or any other characteristic.
We would appreciate acknowledgement if the software is used.





